 Note: We could define mass number of element as the sum of atomic number and quantity of neutrons found in the nucleus. So, we can say that the atomic number of helium is two. So, we saw that the atomic number is the quantity of proton found in the nucleus. We know that the number of protons in Helium is two and the number of electrons in Helium is two. The electrons found in per shell of helium are $2$. We can say that electronic configuration of helium is $1$. In an uncharged molecule, the atomic number is additionally equivalent to the quantity of electrons. It is non-distinguishable from the charge number of the core. The atomic number remarkably recognizes a chemical component. We can define the atomic number or proton number of a compound component as the quantity of protons found in the core of each atom of that component. We know that the physical state of helium is gas. We should know that helium belongs to the s-block and it is seen colorless. We should know that helium is a Group $18$ element and it belongs to period $1$. There are no known compounds that contain helium, although attempts are being made to produce helium diflouride (HeF 2).Hint: We have to know that a chemical element that is represented by symbol is He is helium. Helium is an inert gas and does not easily combine with other elements. The Department of Energy's Jefferson Lab uses large amounts of liquid helium to operate its superconductive electron accelerator. Liquid helium is an important cryogenic material and is used to study superconductivity and to create superconductive magnets. Helium is combined with oxygen to create a nitrogen free atmosphere for deep sea divers so that they will not suffer from a condition known as nitrogen narcosis. It is used as an inert shield for arc welding, to pressurize the fuel tanks of liquid fueled rockets and in supersonic windtunnels. Helium gas is used to inflate blimps, scientific balloons and party balloons. 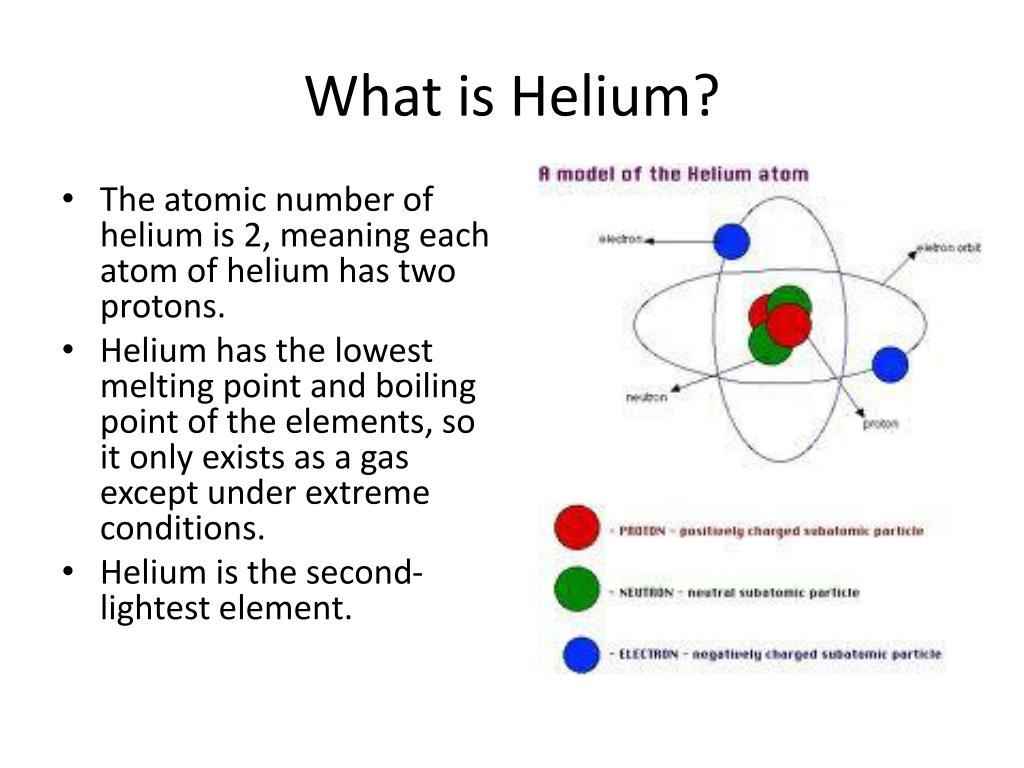
Helium is commercially recovered from natural gas deposits, mostly from Texas, Oklahoma and Kansas. This newly formed helium can eventually work its way to the atmosphere through cracks in the crust. An alpha particle can become a helium atom once it captures two electrons from its surroundings. Alpha decay, one type of radioactive decay, produces particles called alpha particles. The earth's atmospheric helium is replaced by the decay of radioactive elements in the earth's crust. This trace amount of helium is not gravitationally bound to the earth and is constantly lost to space. Helium makes up about 0.0005% of the earth's atmosphere. Two Swedish chemists, Nils Langlet and Per Theodor Cleve, independently found helium in clevite at about the same time as Ramsay. He then sent a sample of these gases to two scientists, Lockyer and Sir William Crookes, who were able to identify the helium within it. He exposed the clevite to mineral acids and collected the gases that were produced. Sir William Ramsay, a Scottish chemist, conducted an experiment with a mineral containing uranium called clevite. The hunt to find helium on earth ended in 1895. 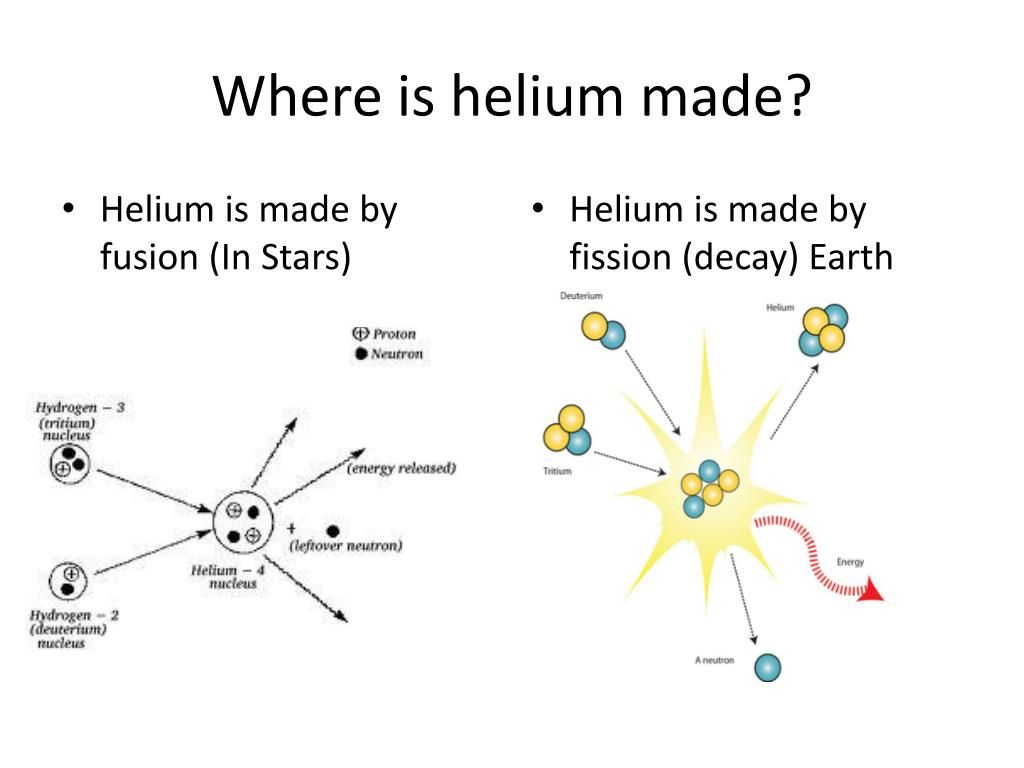
This unknown element was named helium by Lockyer. 
It was hypothesized that a new element on the sun was responsible for this mysterious yellow emission. Sir Norman Lockyer, an English astronomer, realized that this line, with a wavelength of 587.49 nanometers, could not be produced by any element known at the time. Pierre-Jules-César Janssen, a French astronomer, noticed a yellow line in the sun's spectrum while studying a total solar eclipse in 1868. Helium, the second most abundant element in the universe, was discovered on the sun before it was found on the earth. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
